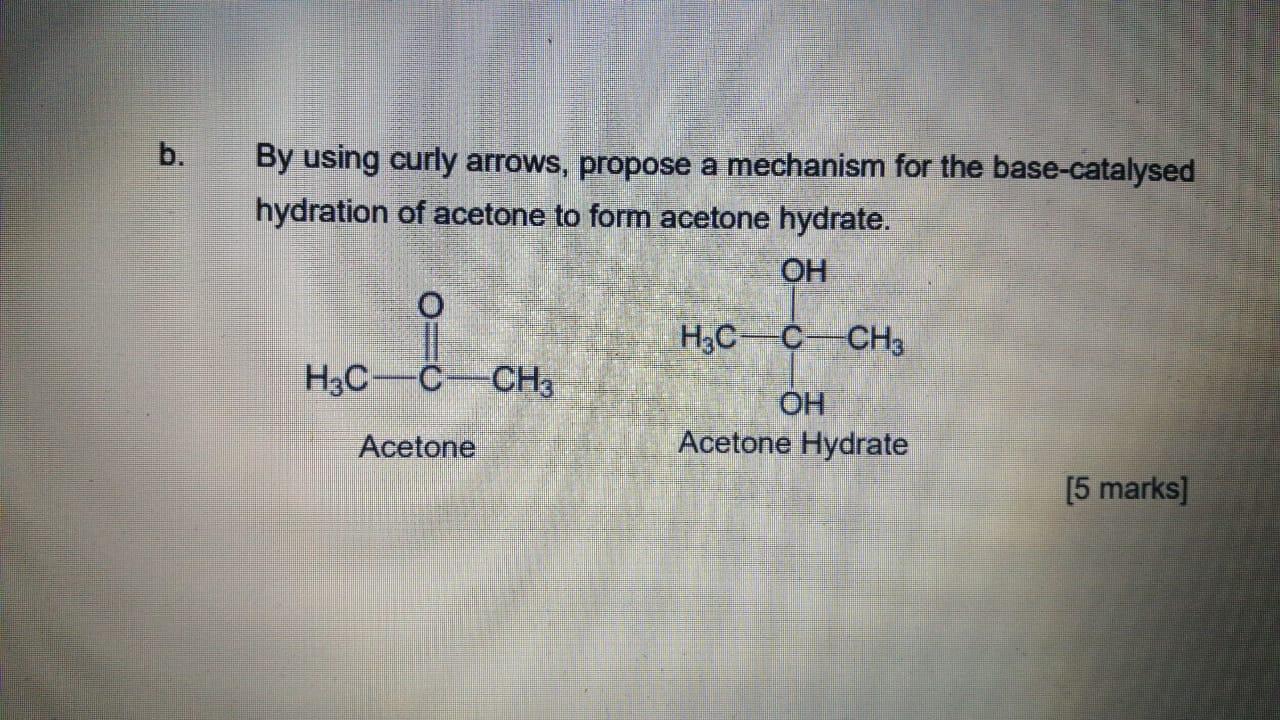
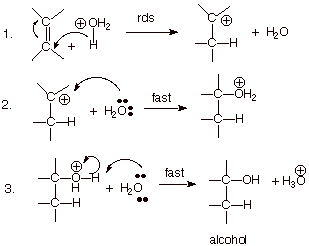
What type of reaction is the reaction between acetone and acid hydronium to yield acetone hydrate? - Quora

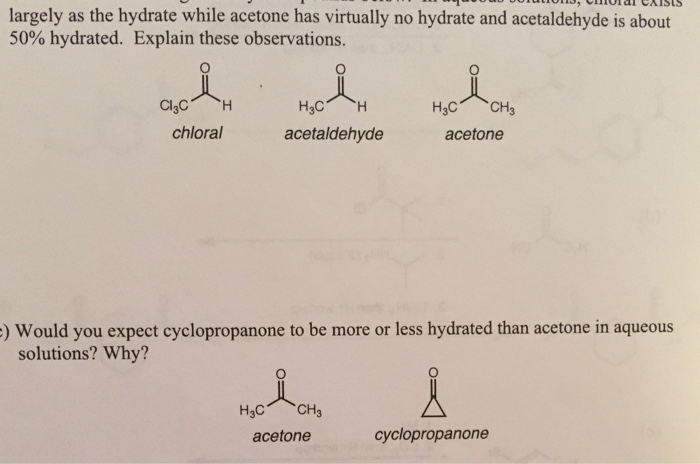
SOLVED:1) In water , approximately 50% of acetaldehyde is present as the hydrate Fore acetone, less than 9 is hydrated. Provide an explanation for this. (5 points) HO OH HO OH Acetaldehyde

Assumed reaction path of the acetone oxime. The starting point of the... | Download Scientific Diagram
![PDF] Phase Equilibrium Data of Binary Hydrate in the System Hydrogen + Acetone + Water † | Semantic Scholar PDF] Phase Equilibrium Data of Binary Hydrate in the System Hydrogen + Acetone + Water † | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/2564829833c04f1098110e9a7be0f8a9f94beaaa/2-Figure1-1.png)
PDF] Phase Equilibrium Data of Binary Hydrate in the System Hydrogen + Acetone + Water † | Semantic Scholar

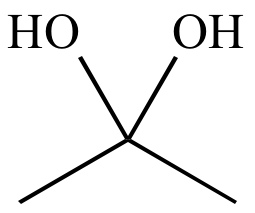
The equilibrium constant for hydration of acetone is 2 times 10^-3 which translates into a small percent conversion to the following hydrate. (Image) Is it possible to expect the equilibrium constan | Study.com

Assumed reaction path of the acetone oxime. The starting point of the... | Download Scientific Diagram