Too many targets, not enough patients: rethinking neuroblastoma clinical trials | Nature Reviews Cancer

TRANSATLANTIC CLINICAL TRIAL FOR CHILDREN WITH NEUROBLASTOMA IS OPEN FOR ENROLLMENT IN THE U.K. - SOON TO OPEN IN THE U.S. AND GERMANY - Band of Parents

CCF on Twitter: "Around 40 children are diagnosed with Neuroblastoma each year. Our funding of the SIOPEN Neuroblastoma Clinical Trials at #ANZCHOG will help identify better therapies and give hope to these

Intellasia East Asia News - Aptorum Group Announces Completion of Phase I Clinical Trials for ALS-4 and SACT-1, Small Molecule Drugs Targeted for Infections Caused by Staphylococcus Aureus and Neuroblastoma

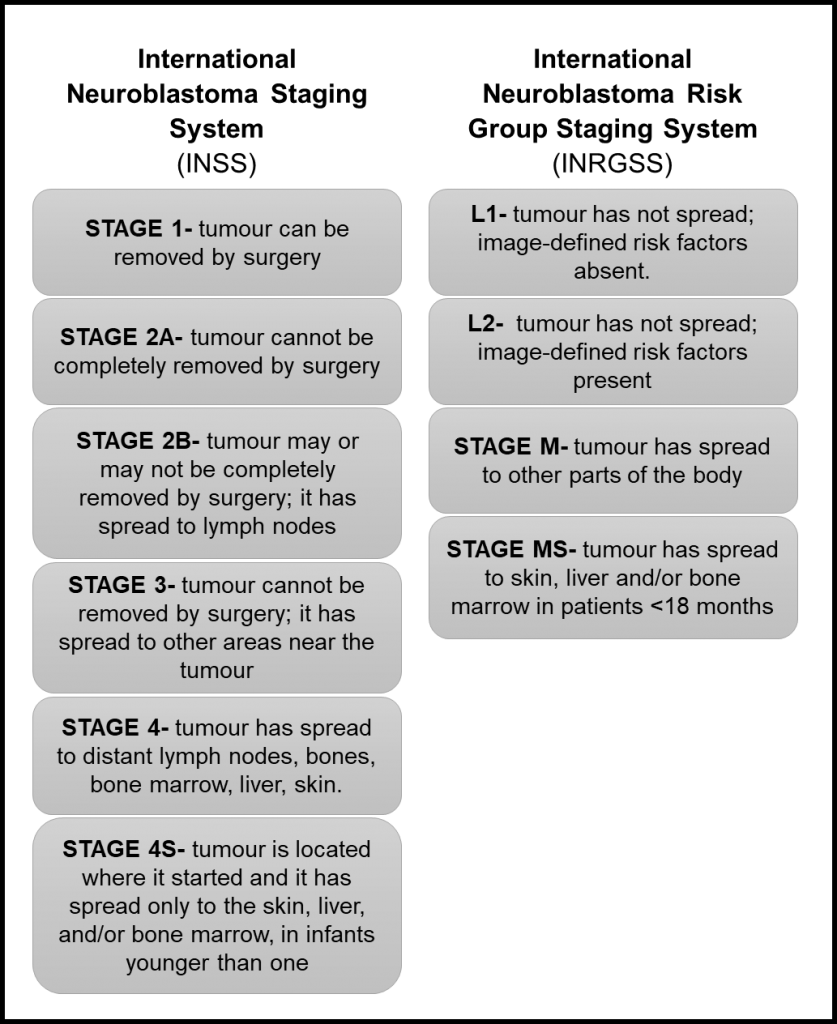
Results of a prospective clinical trial JN‐L‐10 using image‐defined risk factors to inform surgical decisions for children with low‐risk neuroblastoma disease: A report from the Japan Children's Cancer Group Neuroblastoma Committee -

Accelerating drug development for neuroblastoma: Summary of the Second Neuroblastoma Drug Development Strategy forum from Innovative Therapies for Children with Cancer and International Society of Paediatric Oncology Europe Neuroblastoma - European Journal